Effective treatments for a fuller head of hair
See results in as little as 3 months
100% online process
Prescription-strength treatments
Clinically proven ingredients



Spray, serum, and oral meds available
Free shipping in discreet packaging
Unlimited messaging with a provider
Hair growth treatments
3-in-1 Topical Spray
Oral finasteride
Generic Propecia
Oral minoxidil
Generic Loniten
Topical minoxidil
Generic Rogaine
Roman Hair Solution Rx is a compounded prescription medication formulated with finasteride, minoxidil, and tretinoin, applied topically.
Get to know your options
How do these different hair loss treatments work?
We offer both oral and topical treatment options that contain both finasteride and/or minoxidil, two ingredients that are clinically proven to treat hair loss.
Finasteride works to slow, stop, or in some cases even reverse hair loss by blocking the conversion of testosterone to DHT, a hormone that causes male pattern baldness. We offer finasteride in its oral form (generic Propecia) and it’s also available in our 3-in-1 topical spray, Roman Hair Solution Rx.
Minoxidil is thought to help increase blood flow to the hair follicle, helping more nutrients to get to the follicle, which can support hair growth. It’s also thought to extend the growth phase and increase the size of hair follicles. We offer minoxidil in 3 forms; 5% strength topical solution (what you can buy over the counter), 6% strength (as part of our 3-in-1 topical spray), and oral minoxidil, which providers on the Ro platform can prescribe off-label, at their discretion.
Can male pattern baldness actually be treated?
Male pattern baldness occurs when hair follicles undergo a process called follicular miniaturization. This is when the longer, darker hairs that once grew on the scalp get replaced by thinner, shorter, less pigmented hairs. This process occurs due to a hormone called dihydrotestosterone (DHT), which is derived from testosterone.
The sooner you act, the better chance you have at slowing, stopping, or even reversing hair loss. Early on, it is still possible to stop (or even reverse) hair loss. However, at a certain point, the hair follicle also loses attachment to the small muscles in the skin (called the arrector pili muscles). When this happens, hair loss is permanent.
What treatments are proven to slow further hair loss?
In clinical trials, finasteride prevented further hair loss in 83% of men who took it for 2 years and 66% of men even had some hair regrowth. Topical finasteride has also been shown in studies to reduce hair loss in as little as 90 days of treatment.
What treatments are proven to regrow hair?
In a double-blind, placebo-controlled trial of men 18-49 years old, 71% of men using 5% topical minoxidil for 16 weeks. In clinical trials, finasteride prevented further hair loss in 83% of men who took it for 2 years and 66% of men even had some hair regrowth. While the 3-in-1 topical spray is a compounded treatment that hasn’t undergone any clinical testing, it contains two ingredients that are each clinically shown to regrow hair.
Real members, real results
These members received free treatment in exchange for their testimonials.
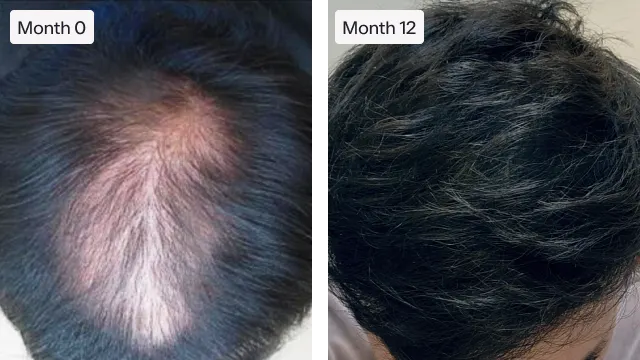
“Very happy with my experience with this company, from the ease of their website to their pricing.”
Yun Jung
Ro member
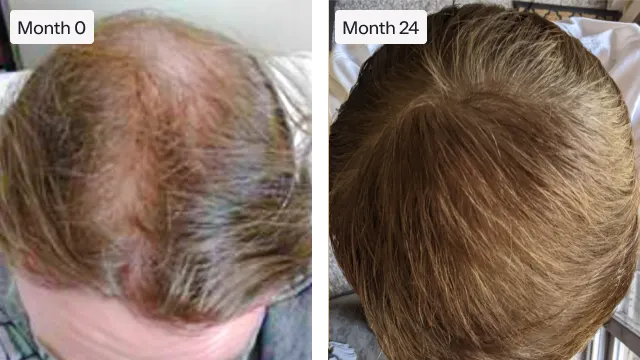
"My hair is [fuller] than it’s been in over a decade, and the bald[ing] spot I was so worried about has shrunk by almost half."
Julian
Ro member
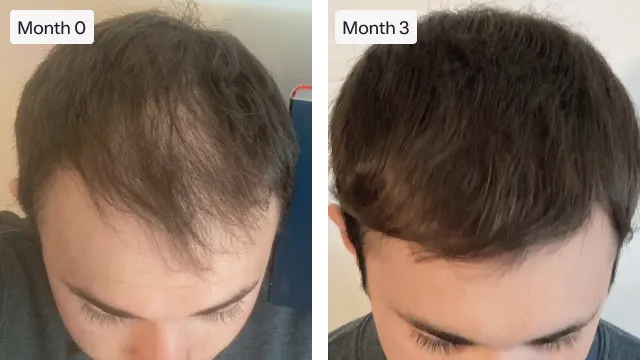
"Roman helped me to regrow my hair and regain my confidence. I used to wear a lot of hats, but now I prefer not wearing one."
Josiah
Ro member
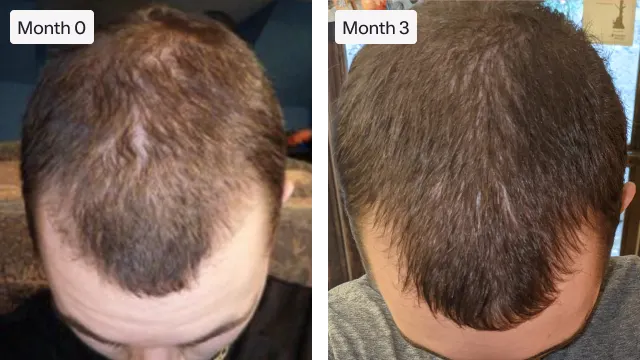
"Roman has helped me regrow and thicken my hair. I’m excited to see more improvement."
Jacob
Ro member

"I feel great about my hair and love that I am more confident. I can still feel young and have a full head of hair."
Cole
Ro member
Join 185,000+ members who treated hair loss with Roman
These members received free treatment in exchange for their testimonials.
Prescription-strength treatments
Clinically proven ingredients

Veljean
Ro member
“Within a few months I saw results. Now my hair is back!”

Gordon B
Ro member
Getting my medicine from Ro has been so easy and convenient. I know that I'm going to have the meds that I need to feel like myself.

Rob
Ro member
“Everything has been fantastic! It’s part of my daily routine now”
Treatment timeline
In 1–2 months
Save it
You might not see a visible change in the first two months, though some people may shed more fine hairs than before. Not to worry—this is temporary and normal.

In 3–6 months
Slow it
You might notice that hair loss has slowed, stopped, or in some cases, even reversed. Remember to take a photo to track your progress.
In 6 months and beyond
Show it off
By now, hair loss may have considerably slowed or stopped, and some people may see signs of regrowth (usually at the crown of the head). To maintain results and increase the chance of regrowth, be sure to continue with consistent daily treatment.
How treating hair loss with Roman works
01
Answer a few questions about your health
After your free online visit, a US-licensed healthcare provider will determine which treatment is best suited for you, then follow up via secure messaging.
02
Get it shipped for free
If treatment is prescribed, you’ll get it shipped fast, free, and in discreet packaging.
03
Get free followups for ongoing care
Have questions or want a follow-up? We offer free, unlimited follow-ups with your provider, so there’s always someone there to help you out. Of course, you should continue to receive regular in-person care in addition to using Roman.
Read up on hair loss
7 min read
8 signs of balding and how to stop it
6 min read
Oral minoxidil: does it work?
5 min read
What happens if you stop taking finasteride?
5 min read
How long does minoxidil take to work?